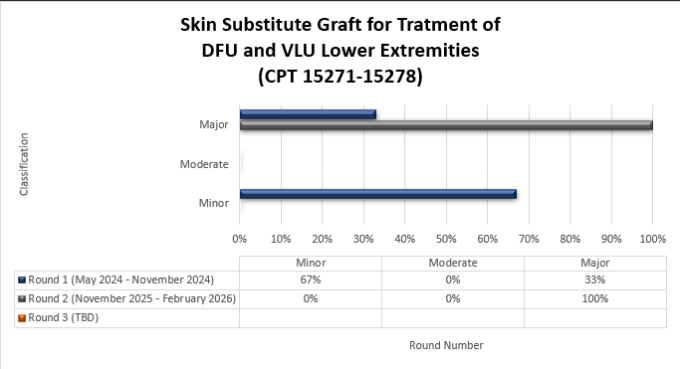
Skin substitute graft for treatment of DFU and VLU lower extremities (15271-15278)
Top denial / partial denial reasons and high-level results are listed below from each round of skin substitutes TPE reviews that have been conducted thus far by Medical Review. If you have questions about your individual results, please contact the nurse reviewer assigned to your review for additional information. Additional rounds of review will be utilized when the targeted topic demonstrates a continued need for review with newly identified providers.
Top denial / partial denial reasons
The most common reasons for denial or partial denials are the following:
- Medical necessity – The documentation submitted does not support medical necessity as listed in coverage requirements.
- Insufficient documentation – Insufficient documentation was provided to support the services as billed to Medicare. Medical Review makes multiple attempts to correct these error types before completion of the review. Below are the following denial reasons for insufficient documentation that we were not able to resolve:
- Documentation submitted lacked the key elements to support the level of service billed.
- Documentation submitted does not support the anatomical area treated.
- Documentation submitted was incomplete and/or insufficient.
- Documentation lacked the initial wound assessment and measurements and the start date of the wound.
- Documentation submitted does not support incident-to-criteria was not met.
- Documentation submitted does not support medical necessity as listed in the coverage requirements in the LCD.
Round results